Education
Back to the Basics of Soil pH

Soil pH is the foundation of soil fertility
Soil pH is a master variable in the soil. It makes everything in the soil, whether biological, chemical or even physical in some instances work together to create a hospitable environment for plants to grow. Managing soil pH is a critical part of a soil fertility plan and can sometimes be overlooked with N, P and K receiving all the attention.
In the video below, I explain why managing soil pH is important and how N, P and K are affected by soil pH.
Managing soil pH improves nutrient availability and yield
In crop production, we shoot for soil pH to be between 6 and 7. Some crops are more sensitive than others to soil pH. The table below shows the relative yield of selected crops at different pH levels. This tells us that at a soil pH of 5.7, corn yield is at 83% of it’s potential.
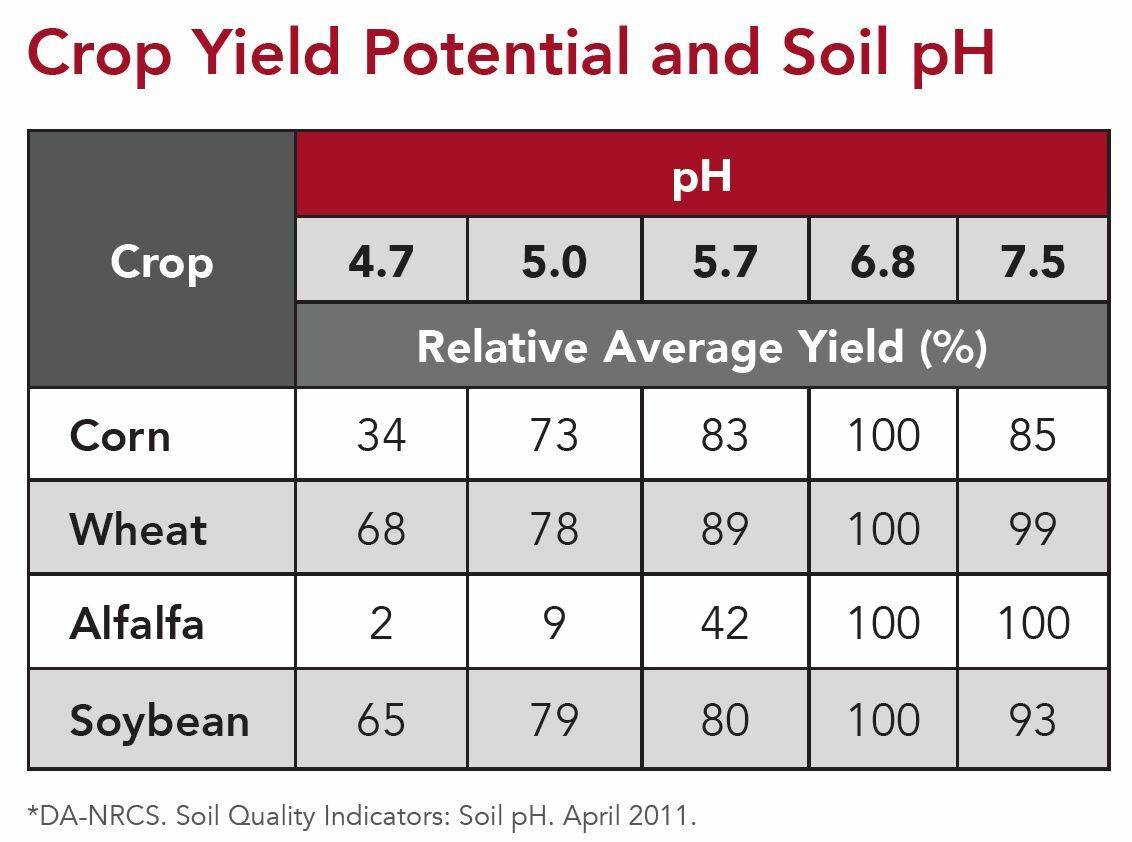
The reason crop yield is impacted by soil pH is largely due to nutrient availability. Phosphorus is the nutrient most impacted by soil pH. At high pH levels, calcium and phosphorus tend to bind up with each other. At low pH, aluminum and iron bind up phosphorus and limit the nutrient’s availability.
While phosphorus is most impacted, nitrogen and potassium are also affected by soil pH. The bacteria responsible for the nitrification process are hindered at low pH, which decreases nitrate availability and forces plants to utilize ammonium, lowering nitrogen use efficiency. Potassium availability is decreased when hydrogen cations from acidification build up, reducing the potential for potassium cations to be held in soil.
Farmers have a substantial investment in their fertility program, so it’s important to make sure soil pH is at a level to put the investment to work.
In addition to nutrient availability, soil pH can also affect herbicide performance and a plant’s genetic yield potential.
