Education
Solubility Differences Among Sulfur Fertilizers

Sulfur is mobile
Sulfur is a mobile nutrient in the soil, which is why the rate at which a fertilizer releases sulfate matters. We’ve completed replicated laboratory trials to better understand sulfur release rates of some of the most common sulfur fertilizers.
Check out the video below where I get into the details of the research we conducted and what we learned.
Release rates of common sulfur fertilizers
To evaluate the rate release, we set up shallow soil cores to see how sulfate was released in the top few inches of soil where plants take up a majority of their nutrients before it leaches lower into the soil. We placed several sulfur fertilizers on top of the soil cores then applied Iowa’s average rainfall through the course of the growing season. Sulfur fertilizers included in the experiment were:
- Ammonium sulfate (AMS)
- Co-granulated MAP + AMS + elemental sulfur
- SO4
- Pelletized synthetic gypsum
- Elemental sulfur
Results were analyzed by Midwest Laboratories and are summarized in this graph.
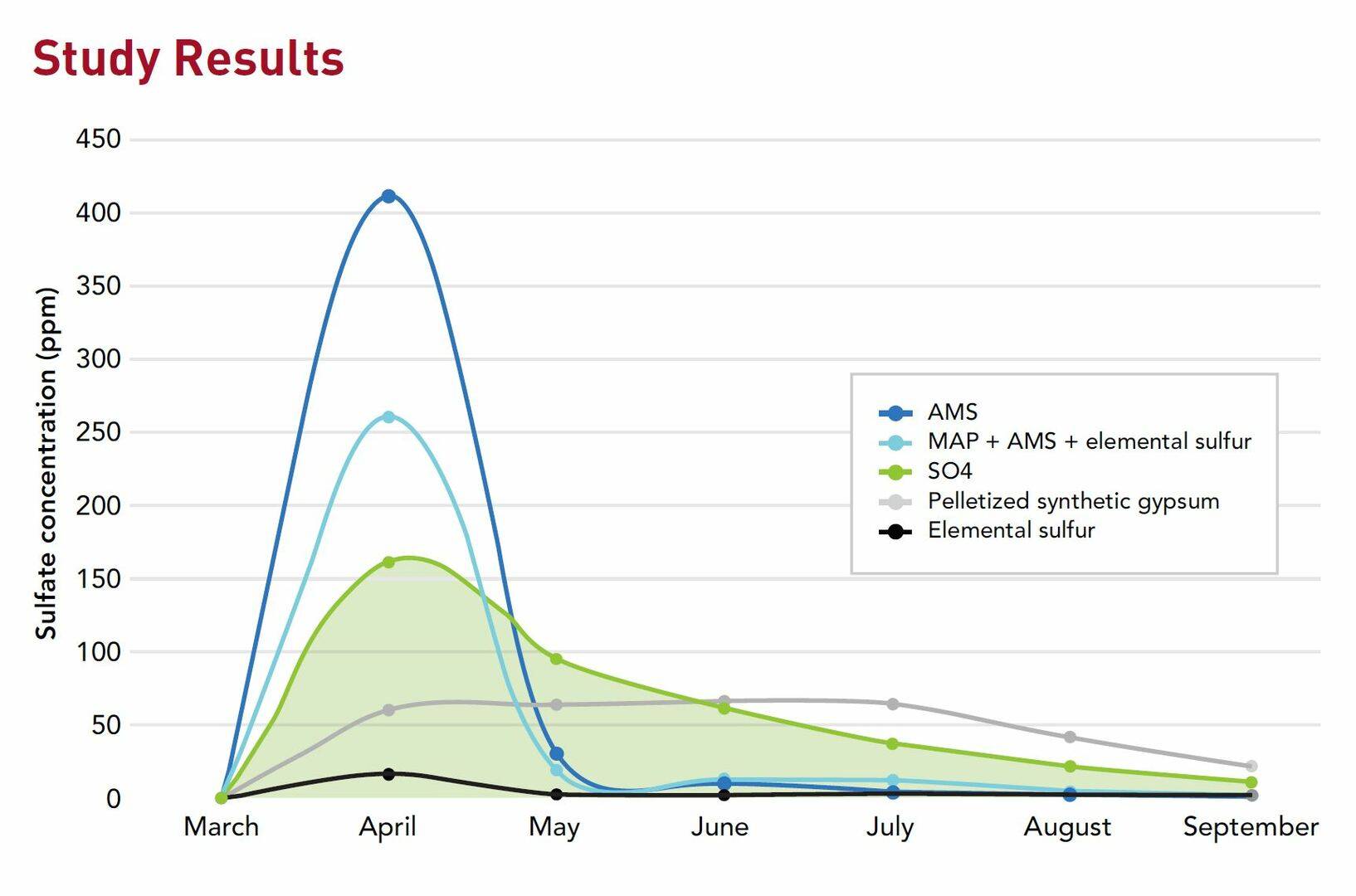
As you can see, AMS has an extreme initial release because it has a very high chemical solubility. This high rate of release makes AMS prone to leaching, especially in wet years.
On the other end of the spectrum is elemental sulfur with a very low release rate, which is because of the oxidation process required to convert the elemental form into sulfate before it can get released into the soil solution.
SO4 provides a nice initial release of sulfur followed by a steady supply throughout the growing season. About 50% of sulfur uptake is needed in corn and soybeans after reproduction is initiated, so availability throughout the growing season is important.
Download the full research brief on sulfur sources and rate of release.
The solubility of sulfur fertilizes varies widely, so it’s worth the time to evaluate and determine which source is the best fit.
